Vast Gene-Expression Map Yields Neurological and Environmental Stress Insights
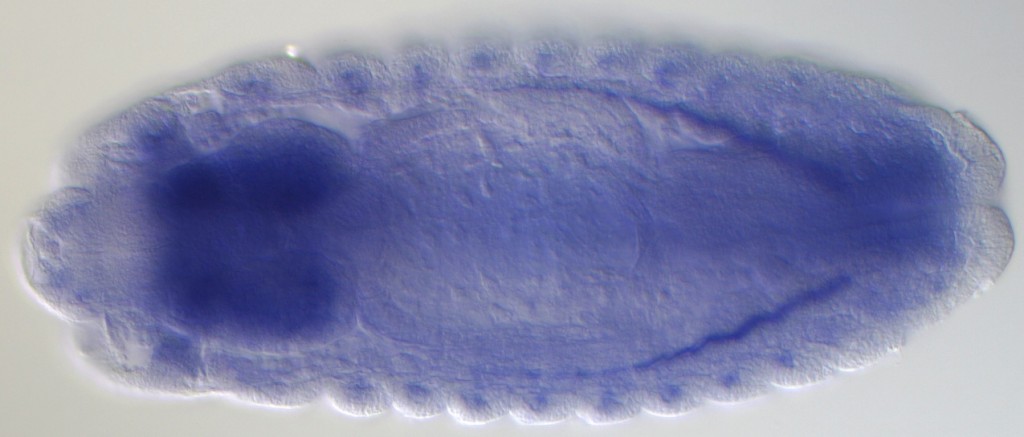
A consortium led by scientists from the U.S. Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) has conducted the largest survey yet of how information encoded in an animal genome is processed in different organs, stages of development, and environmental conditions. Their findings paint a new picture of how genes function in the nervous system and in response to environmental stress.
They report their research this week in the Advance Online Publication of the journal Nature.
The scientists studied the fruit fly, an important model organism in genetics research. Seventy percent of known human disease genes have closely related genes in the fly, yet the fly genome is one-thirtieth the size of ours. Previous fruit fly research has provided insights on cancer, birth defects, addictive behavior, and neurological diseases. It has also advanced our understanding of processes common to all animals such as body patterning and synaptic transmission.
In the latest scientific fruit from the fruit fly, the consortium, led by Susan Celniker of Berkeley Lab’s Life Sciences Division, generated the most comprehensive map of gene expression in any animal to date. Scientists from the University of California at Berkeley, Indiana University at Bloomington, the University of Connecticut Health Center, and several other institutions contributed to the research.
In all organisms, the information encoded in genomes is transcribed into RNA molecules that are either translated into proteins, or utilized to perform functions in the cell. The collection of RNA molecules expressed in a cell is known as its transcriptome, which can be thought of as the “read out” of the genome.
While the genome is essentially the same in every cell in our bodies, the transcriptome is different in each cell type and constantly changing. Cells in cardiac tissue are radically different from those in the gut or the brain, for example.
The transcriptome also changes rapidly in response to environmental challenges. These dynamics in gene expression allow our bodies to adapt to changes such as temperature or exposure to chemicals.
To map the transcriptome, the scientists used deep sequencing technology to generate 1.2 trillion bases of RNA sequence data. They analyzed RNA in 29 fruit fly tissue types, 25 cell lines, and “environmental challenge” scenarios including heat, cold, heavy metal poisoning, and acute exposure to pesticides.
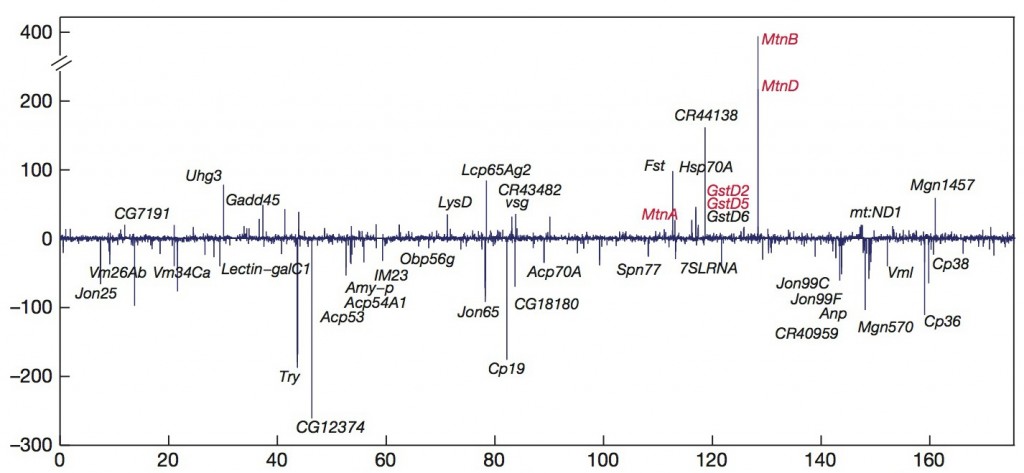
The combination of extremely deep sequencing and a diverse array of tissues and conditions resulted in a full-body map of RNA activity, which revealed new genes and rare RNAs that are expressed in only one tissue type. Among the discoveries are the unexpected complexity and diversity of the RNAs present in tissues of the nervous system, and previously unknown genes implicated in stress response.
In samples of the fly’s nervous system, the scientists found about 100 genes that can encode hundreds or even thousands of different types of proteins. Many of these proteins are made in the developing embryo during the early formation of the nervous system. This hints at a previously unknown source of the complexity of the brain, given that most genes express five or fewer types of transcripts, and half encode just one protein.
“Our study indicates that the total information output of an animal transcriptome is heavily weighted by the needs of the developing nervous system,” says Ben Brown, a Berkeley Lab staff scientist in the Life Sciences Division who led the data analysis team.
The scientists also discovered a much broader response to stress than previously recognized. Exposure to heavy metals like cadmium resulted in the activation of known stress-response pathways that prevent damage to DNA and proteins. It also revealed several new genes of completely unknown function.
“To better understand how cells fight stress, we have to figure out what these mysterious genes do,” says Celniker.
The research was funded by the National Human Genome Research Institute modENCODE Project.
Other institutions involved in this research include the Sloan-Kettering Institute, Japan’s RIKEN Yokohama Institute, Cold Spring Harbor Laboratory, and Harvard Medical School.
###
Lawrence Berkeley National Laboratory addresses the world’s most urgent scientific challenges by advancing sustainable energy, protecting human health, creating new materials, and revealing the origin and fate of the universe. Founded in 1931, Berkeley Lab’s scientific expertise has been recognized with 13 Nobel prizes. The University of California manages Berkeley Lab for the U.S. Department of Energy’s Office of Science. For more, visit www.lbl.gov.
Additional information:
- The Nature paper, “Diversity and dynamics of the Drosophila transcriptome,” appears in Advance Online Publication on March 16.
- A companion paper, “Genome-guided transcript assembly by integrative analysis of RNA sequence data,” appears in Advance Online Publication on Nature Biotechnology’s website on March 16. It describes the methodology behind the Nature paper.
