Pacific Rockfish and the Trade-Offs of a Longer Life

Rockfish is on the menu around the Pacific Rim, for the most part with little regard for the fish’s origin or which of the 137 species is on the plate — it’s typically identified simply as rockfish or, incorrectly, as rock cod or red snapper.
But this seemingly anonymous fish — among the longest-lived vertebrate on Earth — holds clues to the genes that determine lifespan and the pluses and minuses of living longer.
In a study appearing this week in the journal Science, biologists at the University of California, Berkeley, compare the genomes of nearly two-thirds of the known species of rockfish that inhabit coastal waters around the Pacific Ocean and uncover some of the genetic differences that underlie their widely varying lifespans.
Some rockfish, like the colorful calico rockfish (Sebastes dallii), live for little more than a decade, while the most long-lived of the genus Sebastes — the rougheye rockfish (Sebastes aleutianus), which can be found from Japan to the Aleutian Islands — can hang out on the seabed in cold, deep coastal waters for more than 200 years.
Their wide range of lifespans, not to mention differences in size, lifestyle and ecological niche, which scientists call their phenotypes, evolved over a mere 10 million years — one of the most rapid radiations among all fishes.
To uncover the genetic determinants of lifespan in rockfish, the researchers obtained tissue samples — and occasionally tasting samples — from 88 species and sequenced their complete genomes with a state-of-the-art technique known as Pacbio, or SMRT, sequencing.
They found a variety of genes associated with longer lifespan, though some of these genes involve adaptions to living at greater depth and growing larger, both of which are associated with increased lifespan. Among mammals, for example, elephants live longer than rats.
The findings also highlight the trade-offs of a long lifespan, which include smaller populations — something also seen in mammals, with short-lived rats vastly outnumbering long-lived elephants.
“In this study, we identified both the genetic causes and consequences of adaptation to extreme lifespan,” said senior author Peter Sudmant, UC Berkeley assistant professor of integrative biology. “It’s very exciting to be able to look at a group of species and see how their phenotype has been shaped through time and the genetic changes that drive that phenotype, and simultaneously, how that phenotype then feeds back and influences the genetic diversity of that population.”

Sudmant acknowledges that many of the biological pathways he and his team found to be associated with lifespan have been identified before in genetic studies of variation within a single animal species, though this study does implicate several new genes in these pathways. Nevertheless, the natural variation within this one genus of fish that has radiated throughout the Pacific Ocean uniquely encapsulates most of the many genetic factors that influence lifespan.
“You could think of rockfish as sort of the perfect storm. in some ways, both on an individual level — having individual fish able to live for a really long time because of size and depth adaptations — but also just having all these different species that are showing these different trends,” he said. “They’re a perfect set of individuals to look at, where other people just had a single species to look at.”
The study also has implications for understanding human lifespan. Sudmant and his colleagues found that longer-lived species had more immune modulating genes — in particular, a group called butyrophilins — than shorter-lived species. Because the immune system is involved in regulating inflammation, and increased inflammation has been implicated in human aging, the findings point to genes that could be targets of therapeutics to slow age-related damage in the body.
“There is an opportunity here to look in nature and see how natural adaptations have shaped lifespan and to think about how those same sorts of genes are acting in our own bodies,” he said.
Size and habitat explain a lot of lifespan variation in fishes
The researchers looked for DNA variations that were more common in fish with longer lives and found 137 longevity-associated gene variations.
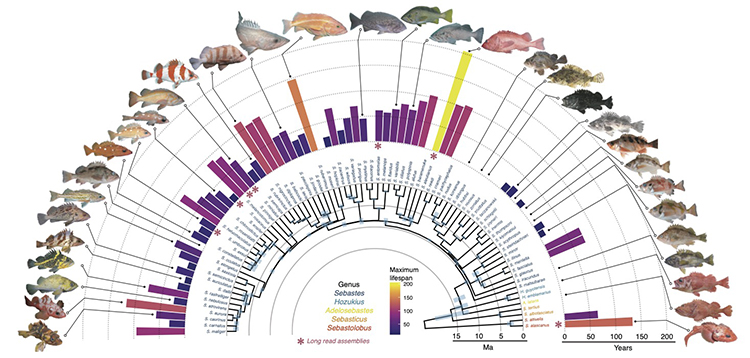
Not all of these have a direct effect on lifespan, however. The researchers took care to separate out the genetic variations that allowed rockfish to adapt to deeper depths and grow to larger size, since those adaptations themselves have the side-effect of increasing lifespan. Deeper, cooler waters slow metabolism, for example, which is associated with a longer lifespan in many animals.
“We can explain 60% of the variation in lifespan just by looking at the size at maturity and the depth at which a fish lives,” Sudmant said. “So, you can predict lifespan with pretty high accuracy just from these factors. This allowed us to identify the genes that allow them to do those things.”
The remainder of the longevity-associated variation primarily involved three types of genes: an enrichment in the number of genes for repairing DNA; variations in many genes that regulate insulin, which has long been known to influence lifespan; and an enrichment for genes that modulate the immune system. More DNA repair genes could help protect against cancer, while more immune genes could help ward off infections, as well as cancer.
“Six different members of the insulin signaling pathway are under selection in these fish,” Sudmant said. “If you look at the textbooks, there’s about nine or 10 core members of the pathway, so the majority of them are under selection in rockfish.”

Essentially, Sudmant said, some rockfish species extended their lifespan simply by adapting to live in deeper, colder waters and increasing their size. The longest-lived species, however, boosted their lifespan even further by tweaking their DNA repair, insulin signaling and immune-modulation genes.
From the 88 rockfish genomes, the researchers also were able to infer what the ancestral rockfish genome looked like and how species evolved from that common ancestor 10 million years ago. With increased lifespan, they found, also came decreased population levels. Some of the longest-lived species survive today in small numbers that rely on very old, but very fertile, females to replenish the population. These big, old, fat, fecund female fish, or BOFFFF, as they are known in fish conservation circles, produce the majority of offspring — sometimes in the millions per year, though with a low survival rate — that seed the next generation.
“In these rockfish, we can actually watch this evolution happening over this 10-million-year time period, and we observe that when some species evolve a short lifespan, their population sizes expand, and when they evolve a long lifespan, their population sizes contract,” he said. “We can see a signature of that in their genomes, in the genetic variation that exists in these species. So, there is a consequence to adapting to long and short life.”
One intriguing finding, he said, is that long-lived species have an excess of certain kinds of DNA mutations — specifically, the conversion of the nucleotide pair CG (cytosine-guanine) to TG (thymine-guanine) — known to accumulate in tumors with aging. Because the oldest females of these long-lived species produce most of the offspring, these unusual genetic alterations are passed along to the rest of the long-lived population.

Sudmant and his lab colleagues are currently involved in similar genome comparisons among bats, primates and other organisms, looking at the genes correlated with lifespan, aging, stress and other phenotypic differences. But the rockfish project was something special, he said.
“Often, in genetics, we’re derided for doing experiments that are fishing expeditions,” he said. “This was both literally and figuratively a fishing expedition.”
UC Berkeley postdoctoral researchers Sree Rohit Raj Kolora and Gregory Owens, now at the University of Victoria in British Columbia, Canada, are co-first authors of the paper. Other co-authors are Juan Manuel Vazquez, Alexander Stubbs, Kamalakar Chatla and professor Doris Bachtrog of UC Berkeley; Conner Jainese, Katelin Seeto, Merit McCrea and Milton Love of UC Santa Barbara; Michael Sandel of the University of West Alabama; Juliana Vianna of the Pontificia Universidad Católica de Chile; and Katherine Maslenikov and James Orr of the University of Washington. The work was funded by an award from the National Institute of General Medical Sciences of the National Institutes of Health (R35GM142916).
RELATED INFORMATION
