How to make space molecules

Complex carbon-based molecules found in meteorites have long been a conundrum for Earth-bound chemists. How could these molecules – a rich zoo of chemical compounds formed from fused rings of carbon and hydrogen – possibly form in the cold vacuum of space?
Chemists at UC Berkeley and Berkeley Lab have now teamed up with scientists at the University of Hawaii at Manoa and Florida International University to clarify one step in this process: the formation of a compound of four interlocking rings – a polycyclic aromatic hydrocarbon, or PAH – called pyrene.
PAHs are found in fossil fuels on Earth and end up as pollutants when burned, but they’re also common in space, especially around giant, carbon-rich stars, and may be the building blocks from which life first developed.
In the past, scientists have conducted lab experiments to recreate the chemical steps leading to polycyclic hydrocarbons with up to 14 carbon atoms.
In a study published this week in the journal Nature Astronomy, the researchers use Berkeley Lab’s Advanced Light Source to simulate conditions around old red giant stars to create pyrene – which has 16 carbon atoms – and related PAHs.
The researchers suspect that building on pyrene through even more ring expansions will produce more complex PAHs, including two-dimensional sheets of carbon like graphene.
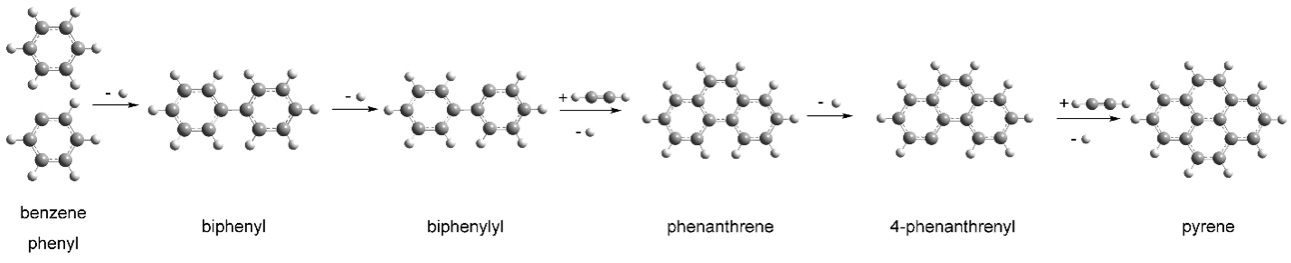
To create pyrene, the researchers first had to create the starting material: a three-ring, 14-carbon PAH called a 4-phenanthrenyl radical. This PAH had been synthesized earlier under star-like conditions, but chemists had gotten no further. Needless to say, this PAH can’t be purchased from a chemical supply house.
“These chemicals are very tedious to synthesize in the laboratory,” said co-author Felix Fischer, an assistant professor of chemistry at UC Berkeley.
At the ALS, they then injected a mix of 4-phenanthrenyl and acetylene – a simple two-carbon molecule – into a microreactor that heated the sample to a high temperature like they might experience near a star.
The gas mixture was then jetted out of the microreactor through a tiny nozzle at supersonic speeds, abruptly stopping the chemical reactions and allowing the scientists to analyze the intermediate reaction products.
Coupled with theoretical calculations of how the reactions should take place, the measurements confirmed the production of pyrene.
“This is how we believe some of the first carbon-based structures evolved in the universe,” said co-author Musahid Ahmed, a scientist in Berkeley Lab’s Chemical Sciences Division.
Future experiments spearheaded by team members at the University of Hawaii will explore what happens when researchers mix hydrocarbon gases in icy conditions and simulate cosmic radiation to see whether that may spark the creation of life-bearing molecules.
“Is this enough of a trigger?” Ahmed said. “There has to be some self-organization and self-assembly involved” to create life forms. “The big question is whether this is something that, inherently, the laws of physics do allow.”
