Fungi helped destroy forests during mass extinction 250 million years ago
The demise of the world’s forests some 250 million years ago likely was accelerated by aggressive tree-killing fungi triggered by global climate change, according to a new study by a University of California, Berkeley, scientist and her Dutch and British colleagues.
The researchers do not rule out the possibility that today’s changing climate could cause a similar increase in fungal diseases that could devastate forests already stressed by warming and drought.
The study, available online today (Aug. 5), will be published in the September 2011 print edition of the journal Geology of the Geological Society of America.
The death of the forests – primarily comprised of conifers, which are distant relatives of today’s pines and firs – was part of the largest extinction of life on Earth, which occurred when today’s continents were part of one supercontinent, Pangaea. The so-called Permian extinction likely was triggered by immense volcanic eruptions in what is now Siberia. The huge amounts of gas and dust thrown into the atmosphere altered global climate, and some 95 percent of marine organisms and 70 percent of land organisms eventually went extinct.
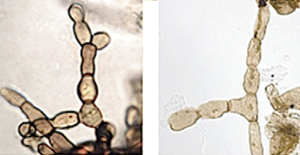
The scientists claim that thread-like or filamentous microfossils commonly preserved in Permian rock are relatives of a group of fungi, Rhizoctonia, that today is known for members that attack and kill plants.
“Modern Rhizoctonia include some of the most ubiquitous plant pathogens, causing root, stem and foliar diseases in a wide variety of plants,” said coauthor Cindy Looy, UC Berkeley assistant professor of integrative biology. “Based on patterns of present-day forest decline, it is likely that fungal disease has been an essential accessory in woodland destabilization, accelerating widespread tree mortality during the end-Permian crisis.”
The conifer forests, which covered the semi-arid equatorial region of Pangaea, were eventually replaced by lycopods – four foot-tall relatives of today’s diminutive club mosses – as well as by seed ferns (pteridosperms). The conifers didn’t recover for another 4 to 5 million years.
Looy and her colleagues – Henk Visscher of the Laboratory of Palaeobotany and Palynology at Utrecht University in the Netherlands and Mark Sephton of the Impacts and Astromaterials Research Centre at Imperial College, London – caution that today’s changing climate could also lead to increased activity of pathogenic soil microbes that could accelerate the death of trees already stressed by higher temperatures and drought.
“Pathogenic fungi are important elements of all forest ecosystems,” said Visscher. “When an entire forest becomes weakened by environmental stress factors, onslaught of damaging fungal diseases can result in large-scale tissue death and tree mortality.”
The researchers dispute the conclusion of other researchers who claim that the thread-like microfossils are the remains of algae. Furthermore, while the researchers previously thought that Reduviasporonites were fungi that took advantage of dying forests, they now believe the fungi actively helped destroy the forests.
“Previously, mass occurrences of Reduviasporonites had been ascribed to wood-rotting fungi living off an excessive abundance of dead wood,” said Looy, a paleobotanist who focuses on pollen and spores as keys to understanding past plant communities. “However, the notion that the microfossils representRhizoctonia-like resting structures suggest a much more active role for fungi in the ecological crisis:”
The researchers’ conclusion comes largely from the fact that they have found living fungi in the genus Rhizoctonia that have a dormant or resting stage during their life cycles in which they look nearly identical to Reduviasporonites.
“One of our problems was that the microfossils didn’t resemble the hyphae of known fungi,” Looy said. “Buta few years ago, we realized that we were looking in the wrong direction; that we should have been looking at fungal resting structures, not normal hyphae.”

Fungi typically spread by means of thread-like hyphae, which can form immense underground networks of mycelia, especially in forests where the fungi live in a symbiotic relationship with tree roots. Each filament is a chain of cells with hard walls made of chitin, the same substance that insects use for their exoskeleton.
When these hyphae branch and intertwine, they may form resting structures known assclerotia. Sclerotia of modern soil-borne fungi such as Rhizoctonia look nearly identical to the disc-shaped structures found among the Reduviasporonites microfossils. Sclerotia are energy storage structures that can help fungi survive extreme conditions.
The team concluded that the loss of trees and the roots that hold soil in place led to severe topsoil erosion, which carried the sclerotia to the sea.
The researchers acknowledge that conifer forests probably suffered from other environmental stresses as a result of the long-term volcanic eruptions, which spewed carbon dioxide and methane into the atmosphere and likely destroyed some of Earth’s protective ozone layer. Nevertheless, they wrote in their paper, “… whatever (the) sequence of events that triggered ecosystem destabilization on land, the aggressiveness of soil-borne pathogenic fungi must have been an integral factor involved in Late Permian forest decline worldwide.”
The work was funded by Utrecht University, Imperial College London and the University of California, Berkeley.
For more information:
- Fungal virulence at the time of the end-Permian biosphere crisis? (Geology, Sept. 2011)
- Cindy Looy’s Web site
