CRISPRing the Microbiome is Just Around the Corner

To date, CRISPR enzymes have been used to edit the genomes of one type of cell at a time: They cut, delete or add genes to a specific kind of cell within a tissue or organ, for example, or to one kind of microbe growing in a test tube.
Now, the University of California, Berkeley, group that invented the CRISPR-Cas9 genome editing technology nearly 10 years ago has found a way to add or modify genes within a community of many different species simultaneously, opening the door to what could be called “community editing.”
While this technology is still exclusively applied in lab settings, it could be used both to edit and to track edited microbes within a natural community, such as in the gut or on the roots of a plant where hundreds or thousands of different microbes congregate. Such tracking becomes necessary as scientists talk about genetically altering microbial populations: inserting genes into microbes in the gut to fix digestive problems, for example, or altering the microbial environment of crops to make them more resilient to pests.
Without a way to track the gene insertions — using a barcode, in this case — such inserted genes could end up anywhere, since microbes routinely share genes among themselves.
“Breaking and changing DNA within isolated microorganisms has been essential to understanding what that DNA does,” said UC Berkeley postdoctoral fellow Benjamin Rubin. “This work helps bring that fundamental approach to microbial communities, which are much more representative of how these microbes live and function in nature.”
While the ability to “shotgun” edit many types of cells or microbes at once could be useful in current industry-scale systems — bioreactors for culturing cells in bulk, for example, the more immediate application may be as a tool in understanding the structure of complex communities of bacteria, archaea and fungi, and gene flow within these diverse populations.
“Eventually, we may be able to eliminate genes that cause sickness in your gut bacteria or make plants more efficient by engineering their microbial partners,” said postdoctoral fellow Brady Cress. “But likely, before we do that, this approach will give us a better understanding of how microbes function within a community.”
Rubin and Cress — both in the lab of CRISPR-Cas9 inventor Jennifer Doudna — and Spencer Diamond, a project scientist in the Innovative Genomics Institute (IGI), are co-first authors of a paper describing the technique that appeared today (Dec. 6) in the journal Nature Microbiology.
From censusing to editing
Diamond works in the laboratory of Jill Banfield, a geomicrobiologist who pioneered the field of community sequencing, or metagenomics: shotgun sequencing all the DNA in a complex community of microbes and assembling this DNA into the full genomes of all these organisms, some of which likely have never been seen before and many of which are impossible to grow in a lab dish.
Metagenomic sequencing has advanced immensely in the past 15 years. In 2019, Diamond assembled 10,000 individual genomes of nearly 800 microbial species from soil samples collected from a grassland meadow in Northern California.
But he compares this to taking a population census: It provides unparalleled information about which microbes are present in which proportions, and which functions those microbes could perform within the community. And it allows you to infer complicated interactions among the organisms and how they may work together to achieve important ecosystem benefits, such as fixing nitrogen. But these observations are only hypotheses; new methods are needed to actually test these functions and interactions at a community level, Diamond said.
“There’s this idea of metabolic handoffs — that no individual microbe is performing a huge string of metabolic functions, but for the most part, each individual organism is doing a single step of a process, and that there has to be some hand-off of metabolites between organisms,” he said. “This is the hypothesis, but how do we actually prove this? How do we get to a point where we’re no longer just watching the birds, we actually can make a few manipulations and see what’s going on? This was the genesis of community editing.”
The research team was led by Banfield, UC Berkeley professor of earth and planetary science and of environmental science, policy and management, and Jennifer Doudna, UC Berkeley professor of molecular and cell biology and of chemistry, Howard Hughes Medical Institute investigator and co-winner of the 2020 Nobel Prize in Chemistry for the invention CRISPR-Cas9 genome editing.
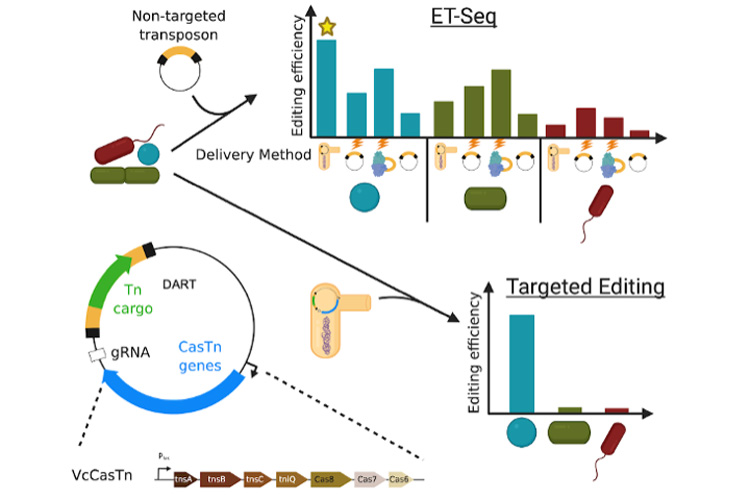
The team first developed an approach to determine which microbes in a community are actually susceptible to gene editing. The screening technique Rubin and Diamond developed, called ET-seq (environmental transformation sequencing), uses as a probe a transposon, or jumping gene, that easily inserts randomly into many microbial genomes. By sequencing the community DNA before and after introducing the transposon, they were able to pinpoint which species of microbes was able to incorporate the transposon gene. The approach was based on techniques developed by co-author Adam Deutschbauer at Lawrence Berkeley National Laboratory. In one experiment involving a community of nine different microbes, they successfully inserted the same transposon into five of them using different transformation methods.
Cress then developed a targeted delivery system called DNA-editing All-in-one RNA-guided CRISPR Cas Transposase (DART) that uses a CRISPR-Cas enzyme similar to CRISPR-Cas9 to home in on a specific DNA sequence and insert a bar-coded transposon.
To test the DART technique with a more realistic microbial community, the researchers took a stool sample from an infant and cultured it to create a stable community composed mostly of 14 different types of microorganisms. They were able to edit individual E. coli strains within that community, targeting genes that have been associated with disease.
The researchers hope to employ the technique to understand artificial, simple communities, such as a plant and its associated microbiome, in a closed box. They can then manipulate community genes within this closed system and track the effect on their bar-coded microbes. These experiments are one aspect of a 10-year program funded by the Department of Energy called m-CAFEs, for Microbial Community Analysis and Functional Evaluation in Soils, which seeks to understand the response of a simple grass microbiome to external changes. Banfield, Doudna, and Deutschbauer are part of the m-CAFEs project.
The research was supported by m-CAFEs (DE-AC02-05CH11231) and the National Institute of General Medical Sciences of the National Institutes of Health (F32GM134694, F32GM131654).
Other co-authors of the paper are Alexander Crits-Christoph, Yue Clare Lou, Adair Borges, Haridha Shivram, Christine He, Michael Xu, Zeyi Zhou, Sara Smith, Rachel Rovinsky, Dylan Smock, Kimberly Tang, Netravathi Krishnappa and Rohan Sachdeva of UC Berkeley; Trenton Owens of Berkeley Lab; and Rodolphe Barrangou of North Carolina State University.
RELATED INFORMATION
