Research Bio
Living cells are not at all well-mixed reaction chambers. Rather, the molecular processes of life occur in dynamic spatial patterns and often operate far from equilibrium. This interplay between spatial organization and the chemical reactions themselves in living systems adds a fascinating dimension to chemistry that is rarely encountered outside of biology. Specific research in Professor Groves' laboratory focuses on how spatial organization influences signal transduction processes at the cell membrane. The research methods combine techniques in optical microscopy and spectroscopy with materials fabrication methods and cell biology. This integrated approach enables the direct observation and physical manipulation of living reaction systems, down to the single molecule level. The conceptual approach is rooted in physics and physical chemistry, with the overarching goal of developing a quantitative and mechanistic understanding biochemical processes in living systems.
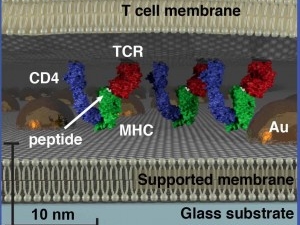
Recent work in the Groves Lab has focused on an emerging type of two-dimensional protein condensation phase transition, which plays important roles in both T cell and growth factor receptor signaling. These systems are studied using a combination of supported membrane microarray reconstitution methods as well as single molecule imaging strategies in live cells. Key questions concern how specific types of phase transition can contribute functional features to biological signaling processes, such as noise filtering, selectivity, and amplification.
Research Expertise and Interest
physical chemistry, intracellular signal transduction mechanisms, physical chemistry of cell membranes, single molecule imaging, stochastic processes
In the News
New Discovery in Living Cell Signaling
Berkeley Lab researchers help find that what was believed to be noise is an important signaling factor. A breakthrough discovery into how living cells process and respond to chemical information could help advance the development of treatments for a large number of cancers and other cellular disorders that have been resistant to therapy.
Berkeley Lab Researchers Demonstrate First Size-based Chromatography Technique for the Study of Living Cells
Using nanodot technology, Berkeley Lab researchers have demonstrated the first size-based form of chromatography that can be used to study the membranes of living cells.
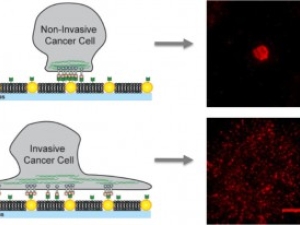
Cancerous Traffic Jams: Biomechanical Factor in Malignancies Identified
Evidence is mounting that the development and spread of cancer, long attributed to gene expression and chemical signaling gone awry, involves a biomechanical component as well.
New Details on the Molecular Machinery of Cancer
Researchers with Lawrence Berkeley National Laboratory (Berkeley Lab) and the University of California (UC) Berkeley have provided important new details into the activation of the epidermal growth factor receptor (EGFR), a cell surface protein that has been strongly linked to a large number of cancers and is a major target of cancer therapies.
A shiny new tool for imaging biomolecules
Berkeley Lab researchers have developed a technique for lacing artificial membranes with billions of gold nanoantennas that can boost optical signals from a protein tens of thousands of times without the protein ever being touched.